 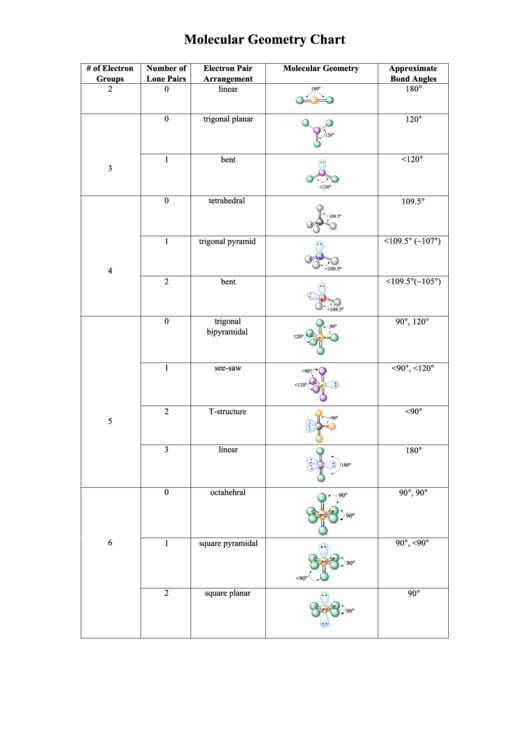
So both Carbon and Hydrogen will share two electrons and form a single bond. If you look at the structure closely, you will realize that Hydrogen can share one electron with the Carbon atom and become stable. Like Hydrogen will have one electron, Carbon will have four electrons, and Nitrogen will have five electrons around its atom like this: Once you have arranged the atoms, start placing the valence electrons around individual atoms. Place the Hydrogen and Nitrogen atoms on both terminal sides of the Carbon like this: And then place the remaining atoms in the structure.Īs Carbon is the least electronegative atom in this molecule, it will take the central position. 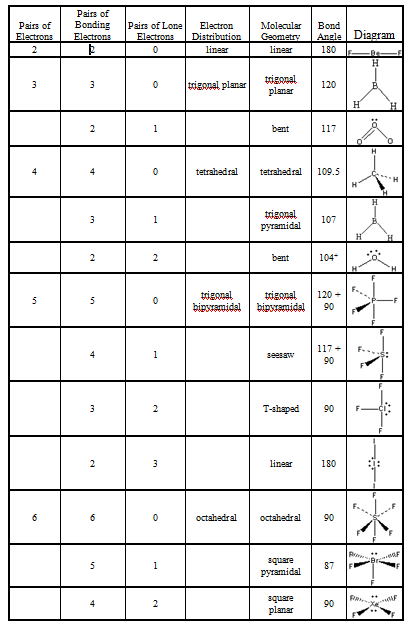
To start with making the Lewis Structure of HCN, we will first determine the central atom. It also aids with understanding the bonds formed in the molecule and the electrons not participating in any bond formation. This structure helps in understanding the arrangement of valence electrons around the atoms in the molecule. Once you get the total number of valence electrons, you can make a Lewis dot structure of HCN. Hence, Hydrogen Cyanide, HCN, has ten valence electrons. of valence electrons in Carbob+ No.of valence electrons in Nitrogen Total number of valence electrons in HCN= No. Whereas Carbon has four valence electrons and Nitrogen has five valence electrons. Hydrogen has one valence electron, and it only needs one more electron to complete its valence shell as it is an exception to the octet rule. This molecule is made up of three different atoms: Hydrogen, Carbon, and Nitrogen. To know the valence electrons of HCN, let us go through the valence electrons of individual atoms in Hydrogen Cyanide. To draw the Lewis dot structure of any molecule, it is essential to know the total number of valence electrons in the structure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
